
ABSTRACTS
The uniqueness of ruminants (Ruminantia) among the even-toed ungulates (Artiodactyla) Part II: Unique anatomical and physiological traits
Jean K. Lightner
In the creationist field of baraminology, one strategy for understanding relationships between creatures is to identify an apobaramin. An apobaramin is a group of organisms that are very distinct from all other organisms, such as chiropterans (bats) which are the only mammals capable of powered flight. In this two-part series we are proposing that ruminants form a clear apobaramin based on both molecular (part 1) and morphologic (here in part 2) characteristics. Many of the unique features of the ruminant stomach are discussed. Osseous headgear is also discussed, as well as its apparent loss from some ruminant families. Further important research necessary to determine if ruminants comprise a single or multiple created kinds is briefly outlined. Ruminants clearly reflect the glory of their Creator, who is all wise and provides for His creatures.
The uniqueness of ruminants (Ruminantia) among the even-toed ungulates (Artiodactyla) Part II: Unique anatomical and physiological traits
Jean K. Lightner
Key Words: Baraminology, ruminant, artiodactyl, rumen anatomy, headgear, cranial appendages, four-chambered stomach
Introduction
One of the purposes of creation research in the field of baraminology is to identify which organisms are truly related due to common ancestry (for an introduction to baraminology, see Friar, 2000). Unlike evolutionists who assume universal common ancestry, creationists affirm the Biblical teaching that organisms were created according to their kinds and endowed with the ability to reproduce and fill the Earth (Genesis 1). It is recognized that organisms change and adapt as they reproduce and fill the Earth, but these changes do not transform one kind of creature into a fundamentally different type of creature. Instead, based on observed examples of evolution, successful changes require pre-existing complexity and pathways that allow for changes that are adaptive, useful agriculturally, or just add beauty and interest. This is the opposite direction of what is necessary to support universal common descent, as the latter would require most changes to build complexity (Spetner, 1998; Anderson and Lightner, 2016).
Identification of created kinds (holobaramins, sometimes popularly called baramins) lays the foundation for a better understanding of biology. When only related organisms are used to infer changes over history, a more realistic understanding of how plants and animals can change can be gleaned. We can discard the magical-sounding stories where massive gene gains must have occurred by naturalistic processes in the past to account for groups of organisms (e.g., insect orders) to come into existence (Wolf and Koonin, 2013; Rosenfeld et al., 2016) and we have a robust explanation for the origin of orphan genes (Neme and Tautz, 2013; Tan, 2015; Yao et al., 2017). A better understanding of how organisms are designed to change may have a significant positive impact on agriculture and medicine.
One way to approach the identification of related organisms (holobarmins) is to identify significant, holistic discontinuity between a group of organisms and all others (Wood, 2003). Such a group is known as an apobaramin, consisting of one or more baramins, with all members united by characters very distinct from all other organisms. This implies they do not share common ancestry with organisms outside that apobaramin. Once an apobaramin has been identified, it can be further investigated to see if there is significant, holistic discontinuity within the group. Eventually, one should be able to identify which groups are truly related, belonging to the same Biblical kind.
Examples of apparent apobaramins would be bats (order Chiroptera), odd-toed ungulates (order Perissodactyla, which includes horses, tapirs, and rhinos), and even-toed ungulates (order Artiodactyla, which includes pigs and peccaries, camels and alpacas, hippos, and ruminants), as species in each of these orders are very distinct from all other organisms. In part one of this series, molecular data was used to demonstrate that ruminants (suborder Ruminantia) mostly cluster together, and do not cluster with any other group. In other words, molecular data support the hypothesis that ruminants form an apobaramin within a larger apobaramin, Artiodactyla. Here the hypothesis is further supported by an examination of two unique traits of ruminants: the ruminant stomach and osseous cranial appendages (headgear).
The Stomach
A simple (monogastric) stomach, as is found in humans and dogs, consists of a single chamber that enzymatically digests food before it passes on to the small intestine. Many herbivores have a more complex digestive system designed to extract nutrients more efficiently from plant food sources. This is accomplished by fermentation in enlarged organs, wherein an abundance of microorganisms break down chemical bonds which their host is not readily able to. In some herbivores the fermentation takes place after the stomach (hind-gut fermentation), where an enlarged cecum and/or colon serves as a large microbial fermentation vat. This is the design in odd-toed ungulates (e.g., horses and rhinos), and several other apobaramins (rabbits and elephants). In contrast, most even-toed ungulates (artiodactyls) have a complex, multi-chambered stomach to allow for fermentation.
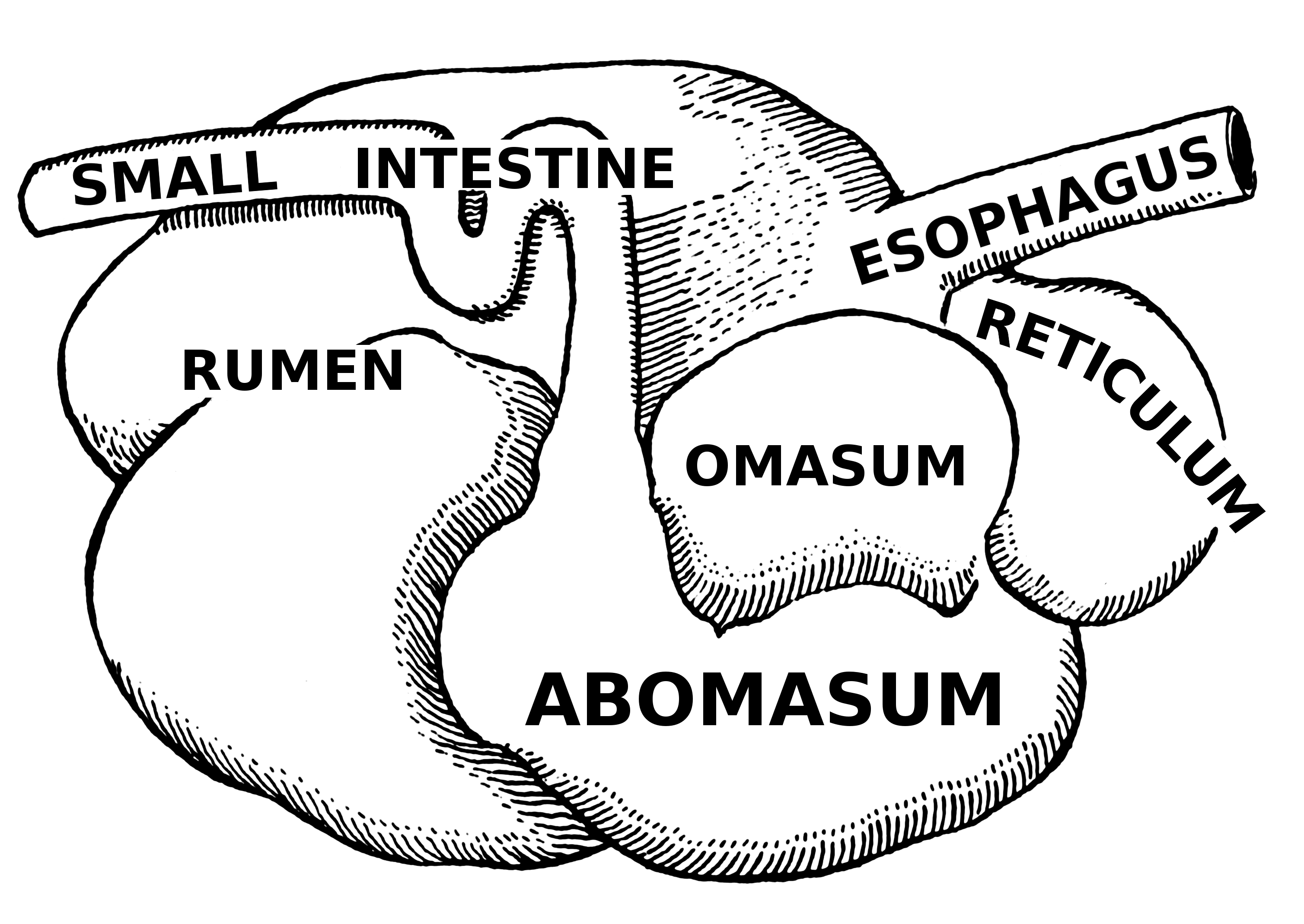
While most other artiodactyls possess two or three-chambered stomachs, ruminants are unique in possessing a very distinctive four-chambered stomach comprised of a rumen, reticulum, omasum, and abomasum (Figure 1) that is highly effective at extracting nutrients from a plant-based diet. Each of these chambers has a distinctive mucosal lining, and the digestive glands are limited to the fourth compartment, the abomasum. The first three compartments are sometimes referred to as the forestomach and the abomasum as the true stomach (Membrive, 2016; Bhamburkar, 2018).
The rumen is partially divided internally by thick muscular bands, called pillars, into a large dorsal and ventral sac, as well as a smaller caudal dorsal and caudal ventral blind sac. The walls of the rumen are lined with papillae which increase surface area and expedite absorption of the products of microbial fermentation, such as short chain fatty acids (SCFAs), B vitamins, and vitamin K (Membrive, 2016; Baldwin and Conner, 2017).
The reticulum is a sac located cranial to the rumen and partially divided from it ventrally by the ruminoreticular fold. Since these two compartments work together and the contents mix relatively freely, they are often referred to together as the ruminoreticulum. The lining of the reticulum includes crests that form four- to six-sided structures, giving it a "honeycomb" appearance. Along the lesser curvature of the reticulum is the reticulo-omasal orifice that serves to promote passage of very small particles to the omasum (Membrive, 2016).
The omasum is generally round or oval. It is lined with semilunar folds that give the interior a book-like or leafy appearance. These folds greatly increase the surface area and allow for considerable water resorption. The result is that the digesta that is passed into the abomasum is much less dilute than what enters the omasum. The abomasum is the glandular stomach, and functions similar to the stomach of monogastrics (Membrive, 2016).
Although it is sometimes claimed that ruminants in the family Tragulidae do not have an omasum, most recent sources recognize them as having a poorly developed omasum (Martin et al., 2001, p. 159; Masters and Flach, 2015). It has been described as a "sac-like transition zone" that has mucosal folds and stratified squamous epithelium (Agungpriyono et al., 1992, 1995). Stratified squamous epithelium lines the first three compartments in a ruminant stomach; this is a unique defining feature of this apobaramin.
The ruminant stomach can adapt. Experiments on domestic ruminants have shown that diet significantly affects physical and metabolic rumen development (reviewed in Baldwin and Conner, 2017). This not only includes rumen mass, but also development and distribution of papillae. Diets higher in concentrates were associated with increased papillae density and height in both calves and lambs. Similarly, diet-related differences in morphology of rumen papillae were observed between wild and domestic deer (Lin et al., 2011; Mason et al., 2019), and with seasonal variations in reindeer (Mathiesen et al., 2006).
In addition to evidence that diet can influence the development and function of the ruminant stomach (physiologic adaptation), there are species-specific differences that suggest a heritable component is involved in many cases (Lentle et al., 1996; Clauss et al., 2017; Przybylo et al., 2019). This is consistent with God creating ruminants with the ability to adapt through designed genetic pathways, enabling both short- and long-term changes. Yet, even in this adaptation, the unique characteristics of the ruminant stomach are preserved.
Since they also chew their cud, camelids have often been compared to ruminants in terms of digestion. Initially, anatomists tried to describe the chambers of the camelid stomach using the terms used in ruminants, but this created problems because of fundamental differences in stomach morphology. Today, the three chambers of the camelid stomach are either numbered (C1, C2, C3) or referred to as the proximal, middle, and distal chambers (reviewed in Alzola et al, 2004; Bravo, 2015). The differences in stomach anatomy are so profound that evolutionists believe the complex stomachs of each evolved independently from a monogastric ancestor (Bravo, 2015, pp. 592-602; Vater et al., 2021).
It should also be noted that, in addition to the lack of head gear and the distinctively different stomach morphology of camelids (Tylopoda) compared to ruminants (Ruminantia), there are a host of other anatomic, physiologic, and behavioral differences between these two suborders. Additionally, the pattern of disease and parasite susceptibility is significantly different between camelids and ruminants (Fowler, 2008, pp. 375-385).
Cranial Appendages (Headgear)
Osseous cranial appendages in extant mammals are unique to ruminants. Four of the extant ruminant families are characterized by headgear: horns are found in bovids, antlers in cervids, ossicones in giraffids, and pronghorns in antilocaprids. While each have unique features, they are all comprised of a bony core covered with integument; their development is characterized by similar gene expression profiles (Wang et al., 2019a).
Horns are found throughout the family Bovidae, though in many species they only appear in the male. Horn bone growth in the bovid neonate originates from horn buds, which are derived from skin. Bony processes develop in two directions. The bony growth directed towards the skull will fuse with the frontal bone, providing a solid foundation for the adult horn. The bony growth directed outwards is covered by a keratin sheath to form the visible portion of the horn (reviewed in Aldersey et al., 2020).
Antlers are unique to cervids; they are the only completely regenerable organ found in mammals. In most species these cranial appendages occur only in the male. They develop annually as an outgrowth of the frontal bone of the skull, and until fully developed, they are covered by a highly vascular layer of sensitive skin known as velvet. As mating season approaches and testosterone levels rise in the male, the antlers harden, and the velvet dries out. A stag will often rub their antlers against objects to remove the flaking velvet. After mating season, testosterone levels fall, osteoclasts dissolve the bone at the base of the antlers, and they fall off (Masters and Flach, 2015, pp. 611-625; Heckeberg, 2017; Wang et al., 2019a).
Ossicones are the headgear of giraffids. Similar to horns, ossification begins separate from the frontal bone. However, they remain detached from the skull (much like scurs in bovids) until sexual maturity and are covered with skin and hair, rather than a keratinous sheath. In contrast, antilocaprid pronghorns develop similar to antlers, or at least fuse much earlier to the skull than horns. They are forked (hence the name pronghorn) and are covered by a deciduous keratinous sheath (Davis et al., 2011; Heckeberg, 2017; Wang et al., 2019a).
Head gear is a trait that can be lost due to genetic mutation. In domestic cattle, four different genetic variants have been characterized in different breeds with polled individuals. These involve complex mutations (indels and/or duplication) on bovine chromosome 1 (BTA 1) in an intergenic region near the centromere. This region is predicted to be a topologically associating domain (TAD), leading to the hypothesis that the mutations affect surrounding genes and/or non-coding RNAs to produce the polled phenotype (Aldersey et al., 2020).
The Chinese water deer (Hydropotes inermis) is an extant cervid that lacks antlers. Similar to the two ruminant families that lack head gear (Moschidae and Tragulidae), it has long upper canines. Molecular study supports the popular hypothesis that both the Chinese water deer and the family Moschidae, lost headgear. It is suggested this was through independent pseudogenization of the RXFP2 gene (Wang et al., 2019b). This inference that these RXFP2 pseudogenes are causal is based, in part, on the observation that an insertion in the 3' untranslated region of this gene in sheep is often correlated with the polled phenotype (Lühken et al., 2016).
Relationships among Ruminant Families
The six extant ruminant families are divided into two groups, or infraorders. Tragulidae is the sole extant family in Tragulina; the other five families are grouped in Pecora. If one wants to make a case that ruminants are polybaraminic, then the most reasonable division is between the tragulids (chevrotains or mouse deer) and remaining ruminants. This hypothesis is also supported by our molecular investigation of artiodactyls in part 1 of this series where the two tragulids represented in the study did not fall in the large ruminant cluster (Lightner and Cserhati, 2023). However, three of the four families represented in the main ruminant cluster had at least one species outside the cluster. Further, tragulids are notoriously understudied (Fernández and Vrba, 2005). Nevertheless, the hypothesis that tragulids represent a distinct baramin within ruminants warrants further study.
Because members of Tragulina occur lower in the fossil record (Eocene) than pecorans (Miocene for extant families), evolutionists logically view the tragulid omasum as an organ that became more fully developed through an evolutionary line leading to pecorans (horn bearers). There are also no horned tragulids, so evolutionists logically believe that pecoran headgear developed later as well. Some hypothesize that headgear arose once among pecoran ruminants; others suggest it arose up to four different times (Davis et al, 2011; Janis and Theodor, 2014).
For creationists holding to a low Flood/post-Flood boundary (at the Cretaceous-Paleogene, or K/Pg), the development of ruminants post-Flood would likely parallel the secular evolutionary progression. While I recognize that significant adaptation can occur because God designed pathways for creatures to adapt (e.g., high altitude adaptation; troglomorphism), I suspect that it is more likely the omasum began as a complex organ, with the ability to decrease in functionality due to adaptation to new food sources, where appropriate. Also, if tragulids are part of the same holobaramin as other ruminants, I would argue they have had a historical loss of headgear as have several pecoran lineages. This latter hypothesis is unlikely to be investigated by evolutionists because of their starting assumptions.
While the pecoran families have remained fairly consistently defined, relationships within these families, as well as between these families, have been highly controversial (Fernández and Vrba, 2005; Davis et al., 2011; Janis and Theodor, 2014). Several possible explanations for this discordance are considered by evolutionists. One explanation is extensive homoplasy (similar form or function among different lineages) combined with poor character selection, as different morphologic studies based on different characters have given differing results (Janis and Theodor, 2014). It is certainly true that if a character is evolvable, or able to change in response to a change in environment or diet, then it can show up a number of different times in different lineages. This parallelism can lead to incorrect conclusions about how these animals are related.
A second possible explanation for the discordant phylogenies is incomplete lineage sorting. In this case an ancestral character was lost in some lineages, but not in others. This also can create a misleading signal when attempting to understand relationships in a group. Finally, introgression (hybridization that brings in new genetics) between lineages is recognized as a possible source (Chen at al., 2019b). Historically, there have been numerous reports of hybridization between bovids and cervids (McCarthy, 2013). Within-family hybridization could also contribute to the difficulty in resolving intrafamilial relationships.
Since the morphologic differences seem to be mostly a matter of modifying or losing an existing structure, any of these may be possible for explaining the discordant phylogenies. However, creationists need to consider one other explanation. Namely, that the families are clearly defined because they were separately created. This, of course, only addresses the confusion on how the families are related to each other; the confusion within families is more likely to be one, or a combination, of the explanations proffered by the evolutionists.
So, plausible creationist hypotheses include 1) a holobaraminic status of ruminants (where differences in the omasum and headgear were derived from created complexity through modification and/or loss), 2) a polybaraminic status with tragulids representing one holobaramin and pecorans a second, and 3) a polybaraminic status with each extant family representing a separate holobaramin (as tentatively proposed in Lightner, 2012).
Regarding the first hypothesis, the location of the Flood/post-Flood boundary would influence the reasonableness of the hypothesis. Otherwise, there are no obvious traits that could not be explained by created complexity followed by modification and/or loss. Tragulids need to be much better studied to make a strong case for or against the second hypothesis. The third hypothesis leaves un-explained the numerous reports of hybrids between two families (Bovidae and Cervidae) as well as the high degree of similarity in external appearance between them. For example, female deer and antelope can bear a striking resemblance to each other.
Regarding the hybrids between cervids and bovids, it is true that none have been confirmed by karyotype or genetic analysis. Thus, despite the numerous claims of such a cross, with varying degrees of documentation, it is best to consider such hybridization tentative until a genetically confirmed example is found. Clearly, God could have made groups of similar organisms. However, given Adam named the progenitors of these animals, a creationist would be justified in assuming the original progenitors were distinctively different looking, and their offspring would largely retain that distinctiveness.
Summary
In this two-part series, a case has been made for the apobaraminic status of ruminants. In part 1 we demonstrated that most ruminant families cluster together based on the Whole Genome K-mer Sequence (WGKS) method. Further, based on the heat maps, no ruminants clustered with any non-ruminant members of the order Artiodactyla. Here in part 2, I have described two unique anatomical traits: the four-chambered ruminant stomach and osseous cranial appendages. The complexity of this anatomy and associated physiology sets ruminants apart from all other mammals. Since there is no observational evidence that this kind of complexity can evolve, ruminants logically represent a group of animals that do not share ancestry with other extant artiodactyls.
It is possible that ruminants comprise more than one created kind. The most obvious differences are between the headgearless tragulids and the pecorans, most of which bear headgear. It is also possible that the five extant pecoran families represent distinct created kinds. However, if the latter is the case, a number of putative hybrid reports between Bovidae and Cervidae remain unexplained. It is also unclear why such similar kinds would have been created, given how similar some females from different extant families (cf. cervids with the bovid antelopes) appear today. Further study to test these various hypotheses is warranted, and the inclusion of fossil data may help clarify this.
Finally, it is concluded that the ruminant stomach was uniquely created; it has the ability to evolve, or change, in ways that allow ruminants to adapt and fill the Earth, but still retains its distinctive features. Likewise, osseous headgear was created, although it is not clear if the differences between the pecoran families are from changes in an original design, or if the variation was created separately. Nevertheless, these impressive characteristics of ruminants provide a glimpse of God's splendor and glory; He is the Creator who provides for His creatures so they can reproduce and fill the Earth. May we honor and rejoice in Him (Psalm 96).
References
Aldersey, J.E., T.S. Sonstegard, J.L. Williams, and C.D.K. Bottema. 2020. Understanding the effects of the bovine POLLED variants. Animal Genetics 51(2): 166-176.
Anderson, K., and J.K. Lightner. 2016. The challenge of Mount Improbable. Creation Research Society Quarterly 52(4): 310-325.
Agungpriyono, S., Y. Yamamoto, N. Kitamura, J. Yamada, K. Sigit, and T. Yamashita. 1992. Morphological study on the stomach of the lesser mouse deer (Tragulus javanicus) with special reference to the internal surface. The Journal of Veterinary Medicine Science 54(6): 1063-1069.
Agungpriyono, S., J. Yamada, N. Kitamura, K. Sigit, Y. Yamamoto, A. Winarto, and T. Yamashita. 1995. Light microscope studies of the stomach of the lesser mouse deer (Tragulus javanicus). European Journal of Morphology 33(1): 59-70.
Alzola, R.H., M.D. Ghezzi, E.J. Gimeno, M.C. Lupidio, A.N. Castro, and J.A. Rodríguez. 2004. Topography and morphology of the llama (Lama glama) stomach. International Journal of Morphology 22(2): 155-164; https://dx.doi.org/10.4067/S0717-95022004000200010 .
Baldwin, R.L., and E.E. Connor. 2017. Rumen function and development. The Veterinary Clinics of North America: Food Animal Practice 33(3): 427-439.
Bhamburkar, R.V. 2018. Veterinary Anatomy: The Regional Gross Anatomy of the Domestic Animals. New India Publishing Agency, (Pitampura, Delhi) New Delhi, India.
Bravo, P.W. 2015. Chapter 60 - Camelidae. In Miller, R.E., and M.E. Fowler (editors), Fowler's Zoo and Wild Animal Medicine, Volume 8. Elsevier Saunders, St. Louis, MO; https://doi.org/10.1016/B978-1-4557-7397-8.00060-8 .
Clauss, M., J. Fritz, A. Tschuor, U. Braun, J. Hummel, and D. Codron. 2017. Dry matter and digesta particle size gradients along the goat digestive tract on grass and browse diets. Journal of Animal Physiology and Animal Nutrition 101(1): 61-69; https://doi.org/10.1111/jpn.12505 .
Davis, E.B., K.A. Brakora, and A.H. Lee. 2011. Evolution of ruminant headgear: A review. Proceedings of the Royal Society B 278(1720): 2857-2865.
Fernández, M.H., and E.S. Vrba. 2005. A complete estimate of the phylogenetic relationships in Ruminantia: a dated species-level supertree of the extant ruminants. Biological Reviews of the Cambridge Philosophical Society 80(2): 269-302.
Fowler, M.E., and R.E. Miller (editors). 2008. Camelids are not ruminants. In Zoo and Wild Animal Medicine, Sixth Edition (Volume 6). Saunders, Phildelphia, PA; https://doi.org/10.1016/B978-141604047-7.50049-X .
Friar, W. 2000. Baraminology - Classification of created organisms. Creation Research Society Quarterly 37(2): 82-91.
Heckeberg, N.S. 2017. Origination of antlerogenesis. Journal of Morphology 278(2): 182-202.
Janis, C.M., and J.M. Theodor. Cranial and postcranial morphological data in ruminant phylogenetics. Zitteliana B 32(32): 15-31.
Lentle, R.G., I.M. Henderson, and K.J. Stafford. 1996. A multivariate analysis of rumen papillary size in red deer (Cervus elaphus). Canadian Journal of Zoology 74(11): 2089-2094; https://doi.org/10.1139/z96-237 .
Lightner, J.K. 2012. Mammalian Ark kinds. Answers Research Journal 5: 151-204; https://assets.answersingenesis.org/doc/articles/pdf-versions/arj/v5/mammalian-ark-kinds.pdf (accessed December 22, 2020).
Lightner, J.K., and M. Cserhati. 2023. The uniqueness of ruminants (Ruminantia) among the even-toed ungulates (Artiodactyla), Part I: Molecular baraminology studies. Creation Research Society Quarterly 59(3): 142-150.
Lin, M.F., S.L Ang, C.W. Yang, J.T Hsu, and H.T. Wang. 2011. Study on the characteristics of gastrointestinal tract and rumen ecology of Formosan Reeves'. Journal of Applied Animal Research 39(2): 142-146; https://doi.org/10.1080/09712119.2011.565560 .
Lühken, G., S. Krebs, S. Rothammer, J. Küpper, B. Mioč, I. Russ, and I. Medugorac. 2016. The 1.78-kb insertion in the 3'-untranslated region of RXFP2 does not segregate with horn status in sheep breeds with variable horn status. Genetics, Selection, Evolution 48(1): 78. doi: 10.1186/s12711-016-0256-3.
Martin, R.E., R.H. Pine, and A.F. DeBlase. 2001. A Manual of Mammalogy, Third Edition. McGraw-Hill Publishing, Boston, MA.
Mathiesen, S.D., Ø.E. Haga, T. Kaino, and N.J.C. Tyler. 2006. Diet composition, rumen papillation and maintenance of carcass mass in female Norwegian reindeer (Rangifer tarandus tarandus) in winter. Journal of Zoology 251(1): 129-138; https://doi.org/10.1111/j.1469-7998.2000.tb00598.x .
Mason, F., B. Fotschki, A. Di Rosso, and A. Korzekwa, A. 2019. Influence of farming conditions on the rumen of red deer (Cervus elaphus). Animals (Basel) 9(9): 601; https://doi.org/10.3390/ani9090601 .
Masters, N., and E. Flach. 2015. Chapter 62 - Tragulidae, Moschidae, and Cervidae. In R.E. Miller and M.E. Fowler (editors). Fowler's Zoo and Wild Animal Medicine, Volume 8. Elsevier Saunders, St. Louis, MO; https://doi.org/10.1016/B978-1-4557-7397-8.00062-1 .
McCarthy, E.M. 2013. Deer-cow hybrids. Mammalian Hybrids. http://www.macroevolution.net/deer-cow-hybrids.html (accessed December 21, 2020).
Membrive, C.M.B. 2016. Anatomy and Physiology of the Rumen. In D.D. Millen, M. De Beni Arrigoni, and R.D. Lauritano Pacheco (editors). Rumenology. Springer International Publishing, Cham, Switzerland.
Neme, R., and D. Tautz. 2013. Phylogenetic patterns of emergence of new genes support a model of frequent de novo evolution. BMC Genomics 14: 117. doi: 10.1186/1471-2164-14-117.
Przybyło, M., J. Hummel, S. Ortmann, D. Codron, G.M. Kohlschein, D. Kilga, J. Smithyman, U. Przybyło, S. Świerk, S. Hammer, J.M. Hatt, P. Górka, and M. Clauss. 2019. Digesta passage in nondomestic ruminants: Separation mechanisms in 'moose-type' and 'cattle-type' species, and seemingly atypical browsers. Comparative Biochemistry and Physiology. Part A, Molecular & Integrative Physiology 235: 180-192; https://doi.org/10.1016/j.cbpa.2019.06.010 .
Rosenfeld, J.A., J. Foox, and R. DeSalle. 2016. Insect genome content phylogeny and functional annotation of core insect genomes. Molecular Phylogenetics and Evolution 97: 224-232.
Spetner, L.M. 1998. Not by chance: Shattering the modern theory of evolution. The Judaica Press, Brooklyn, NY.
Tan, C.L. 2015. Using taxonomically restricted essential genes to determine whether two organisms can belong to the same family tree. Answers Research Journal 8: 413-435.
Vater, A.L., E. Zandt, and J. Maierl. 2021. The topographic and systematic anatomy of the alpaca stomach. Anatomical Record 304(9): 1999-2013. doi: 10.1002/ar.24588.
Wang, Y., C. Zhang, N. Wang, Z. Li, R. Heller, R. Liu, Y. Zhao, J. Han, X. Pan, Z. Zhang, X. Dai, C. Chen, M. Dou, S. Peng, X. Chen, J. Liu, M. Li, K. Wang, C. Liu, Z. Lin, L. Chen, F. Hao, W. Zhu, C. Song, C. Zhao, C. Zheng, J. Wang, S. Hu, C. Li, H. Yang, L. Jiang, G. Li, M. Liu, T.S. Sonstegard, G. Zhang, Y. Jiang, W. Wang, and Q. Qiu. 2019a. Genetic basis of ruminant headgear and rapid antler regeneration. Science 364(6446): eaav6335. doi: 10.1126/science.aav6335.
Wang, B., L. Chen, and W. Wang. 2019b. Genomic insights into ruminant evolution: From past to future prospects. Zoological Research 40(6): 476-487.
Wolf, Y.I., and E.V. Koonin. 2013. Genome reduction as the dominant mode of evolution. Bioessays 35(9): 829-837.
Wood, T.C., K.P. Wise, R. Sanders, and N. Doran. 2003. A refined baramin concept. Occasional Papers of the Biology Study Group 3: 1-14.
Yao, C., H. Yan, X. Zhang, R. Wang. 2017. A database for orphan genes in Poaceae. Experimental and Therapeutic Medicine 14(4): 2917-2924.
Figure 1: https://en.wikipedia.org/wiki/Ruminant#/media/File:Abomasum_(PSF).png
A stylized drawing of the ruminant stomach.
